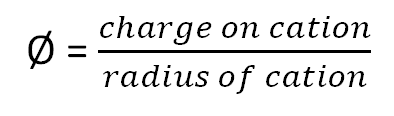Polarizability Formula and Definition
The ratio of the size of an ion to its charge is called charge density. Generally, positive ions have higher charge density and smaller size and negative ions have a large charge but low charge density (except F– and 0-2 ions). The ease with which the charge distribution in a molecule can be distorted by an external electric field is called its polarizability.
We can think of the polarizability of a molecule as a measure of the “squishiness” of its electron cloud: the greater the polarizability of a molecule, the more easily its charge can be distorted to give a momentary dipole.
In general, larger molecules tend to have greater polarizabilities because they have a greater number of electrons and their electrons are further from the nuclei.
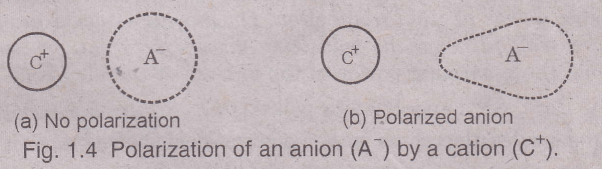
When a cation approaches an anion closely, the net positive charge on the cation tends to attract the electron cloud of the anion, towards itself due to the electrostatic force of attraction existing between them. At the same time, the cation also tends to repel the positively charged nucleus of the anion.
Polarization Power
The combined effect of these two forces is that the electron cloud of the anion no longer remains symmetrical but is bulged or elongated towards the cation. This is called distortion, deformation or polarization of the anion by the cation and the anion is said to be polarized. The ability of a cation to polarize (or distort) a nearby anion is called its polarization ability or polarization power.
The cation is also polarized by the anion, but because the size of a cation is generally smaller than anion, the cation is polarized by the anion to a less extent i.e., the polarization of a cation by an anion is considered almost negligible in most cases.
We say, therefore, that the cations have a strong polarization power and the anions are more susceptible to polarization. Thus we see that when a cation and anion come closer to each other, both ions are polarized, but the polarization produced in anion by cation is appreciable while that produced in cation by anion is negligible.
Fajans’ Rules
Fajans considered the effect which a small, highly charged cation would have on an anion. If the anion were large and “soft” enough, the cation should be capable of polarizing it, and the extreme of this situation would be the cation actually penetrating the anionic electron cloud giving a covalent (shared electron) bond.
Fajans suggested the following rules to estimate the extent to which a cation could polarize an anion and thus induce covalent character.
Following are the rules on which the magnitude of polarizing power of a cation to polarize a nearby anion and the polarizability of an anion depends on:
- Small, highly charged cations will exert a greater effect in polarizing anions than large and /or singly charged cations.
- High charge and large size of the anion will be more strongly or easily polarized by a given cation and hence greater will be the tendency of the anion to form covalent compounds.
- The cation with ns2p6d10 configuration has greater polarizing power than the cation with 8-electron valence shell configuration (ns2p6 configuration) and hence has a greater tendency to form covalent compounds.
Periodic Trends of the Polarizing power of cations
(i) In a period: In moving from left to right in a period, the size of the cations decreases, and the positive charge increases. Both these factors increase the polarizing power of the cations from left to right in a period.
For example, the polarizing power of the cations of the 3rd period, i.e., Na+, Mg2+, and Al3 to polarize cl– anion in NaCl, MgC12 and A1Cl3 molecules increase from Na+ to Al3- a decrease of their size and increase in their positive charge from +1 to +3.
(ii) In a group On descending a group, the magnitude of positive charge remains in the same and hence it is the size of the cation alone which affects the magnitude of the polarizing power of the given cation.
On moving down a group the size of the cations increases and hence the polarizing power of the cation goes on decreasing.
For example, the polarizing power of the cations of the elements of Group IIA Be2+, Mg2+, Ca 2+, Sr2+, and Ba2+ to polarize Cl – ion in BeC12, MgC12, CaC I2, rC12 and BaC12 molecules, decreases with the increase of their size.
Periodic Trends of Polarizability of Anions
- In a Period: In moving from left to right in a period, both negative charge on the anions and their size decrease. Both these factors decrease the polarizability of the anions by given cations from left to right in a period of the periodic table.
For example, the polarizability of c-4, N-3, 0-2, and -F anions (anions of 2nd period) to be polarized by M+ cations in M4C, M3N, M20, and MF molecules decreases from C to – F. Since, both negative charge on these anions and their size .also decrease in the same direction.
- In a Group: On descending a group, the magnitude of negative charge on the anions remains the same and hence it is the size of the anion alone which affects the magnitude of the polarizability of a given anion. On moving down a group the size of the anions increases and hence the polarizability of the anions also goes on increasing.
For example, in the halides of a given cation (e.g. MX type halides) the polarizability of halide ions (anions of the same group) will increase from F–to I-. since the size of these anions also increases in the same direction.
Applications of the concept of Polarization or Polarizability
The polarization power of a cation is usually called ionic potential or charge density and is represented by Ø (phi). It is expressed mathematically as:
- The degree of covalent character in an ionic compound: The larger the value of Ø for a cation, the greater is its polarization power to polarize the electron cloud of an anion, and hence greater is the degree of covalent character in its ionic compound.
- The tendency of a cation towards salvation: The larger the value of ¢ for a cation, the greater is its tendency towards salvation. It is, for this reason, the degree of hydration decreases from Li+ to Cs+
- The tendency of a cation to form complexes: The larger the value of¢ for a cation, the greater is its tendency to form complexes.
- Nature of Oxides: Larger the value of Ø for a cation of an oxide, greater is the polarization. power of the cation and greater will be the covalent character of the metal-to -oxygen bond. The increase in the acidic nature is due to the increase in the covalent character of the metal-to-oxygen.
- Bond energy: Polarization tends to increase the bond.
- Melting points of compounds: With the increase of polarization, the degree of covalent character in an ionic compound also increases and this increased covalent character decreases the melting points of the compound.
- Solubility: With the increase of polarization, the solubility of the compound decreases. It is for this reason that the sulfides are less soluble than the oxides of the same.